Reaction of Concentrated Hydrochloric Acid and Manganese Dioxide
What Is Manganese Dioxide
Manganese dioxide is an amphoteric oxide. It is a very stable black powdery solid at room temperature and can be used as a depolarizer for dry batteries. Physical properties: black amorphous powder, or black orthorhombic crystals. Solubility: Insoluble in water, weak acid, weak base, nitric acid, cold sulfuric acid, and soluble in concentrated hydrochloric acid under heating to produce chlorine gas.
The Physical And Chemical Properties of Manganese Dioxide
1. Physical properties
The appearance of manganese dioxide is black or black brown crystals or amorphous powder. The decomposition temperature is approximately 535°C and the relative density is 5.03. This product is insoluble in water and nitric acid.
2. Chemical properties
Manganese dioxide is an amphoteric oxide. It is a very stable black powdery solid at room temperature and can be used as a depolarizer for dry batteries. It is often used in the laboratory to produce chlorine by its oxidizing property and the action of concentrated HCl.
Concentrated Hydrochloric Acid And Manganese Dioxide React to Produce Hydrogen
Hydrochloric acid is a test reagent often used in chemical experiment operations, and it is also often used in industrial products. Concentrated hydrochloric acid is one of the six strong inorganic bases in industrial production. Hydrochloric acid is generally a colorless liquid. When sprayed into the air, it will cause mist and a choking smell. Manganese dioxide is an amphoteric oxide. Using its reliability at room temperature, it is used in the production of raw materials for rechargeable batteries. Manganese dioxide and concentrated hydrochloric acid are used in the laboratory to produce chlorine.
Manganese dioxide has general functions and main uses. As a battery depolarizer, it produces industrially produced metal catalysts and reducing agents, as well as additives, decolorizers, and de-irons produced in laminated glass industrial production and Tang porcelain industrial production. It is used to produce metal manganese, special aluminum alloys, ferromanganese castings, gas masks and electronic material ferrite cores. In addition, it can also be used for industrial production of vulcanized rubber to improve the viscosity of vulcanized rubber. It can also be used as a metal catalyst in chemical experiment operations. Manganese dioxide is very effective in analytical chemistry. The shapes of manganese dioxide used as metal oxides vary. Because manganese dioxide has several crystal shapes, the chemical equation can be written as MnO2-x(H2O)n, where x is between 0 and 0.5, and n can exceed 0.
Manganese dioxide can be caused by the reaction of potassium permanganate solution and manganese sulfate at different pH. The brown manganese dioxide precipitate is very dry and very active. The most effective organic chemical solvents include aromatic substances, carbon tetrachloride, ethers, tetrahydrofuran and lipids. Used as a metal catalyst when hydrogen peroxide is dissolved to produce oxygen. Used as a metal catalyst when heating potassium chlorate to dissolve oxygen. It produces thermite reaction with hydroxide alumina powder to prepare manganese. As color paste, light yellow laminated glass, etc. React with hot concentrated hydrochloric acid to produce chlorine. It reacts with molten caustic potash in the air to obtain potassium manganate.
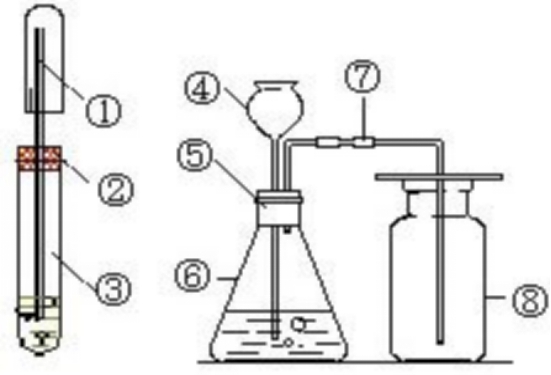
Hydrochloric acid is a key basic chemical raw material and is widely used. The key is to produce a variety of fluorides; obtain a variety of non-ferrous metals in chemical processes; in organic chemistry, textile bleaching, petroleum processing, tannery paper industry, electroplating process arc welding, metal material pickling Chemicals are common acids; in daily life, it can remove rust and scale; in the production of organic chemicals, procaine, thiamine hydrochloride, glucose, etc. cannot be lacked; in the production of animal glue, various The sample dye also has a foothold; in the food industry, it is used to make chicken essence and organically pumped by students; the doctor immediately allows patients with insufficient gastric juice to take extremely dilute hydrochloric acid to cure indigestion; in scientific research and chemical experiment operations It is one of the most common chemicals.
The reaction of concentrated hydrochloric acid and manganese dioxide is MnO2+4HCl (concentrated)=MnCl2+Cl2+2H2O. This method can be used to produce hydrogen.
Luoyang Trunnano Tech Co., Ltd (TRUNNANO) is a professional manganese dioxide with over 12 years experience in chemical products research and development. If you are looking for high quality manganese dioxide, please feel free to contact us and send an inquiry.
Leave a Reply
- A Comprehensive Parameter-Based Analysis of Silicon Carbide Industrial Ceramics: Types, Properties, and Applications
- Sodium Silicate, Potassium Silicate, Lithium Silicate, Silica Sol and Aerogel: Comparison of Properties and Application Analysis
- Unveiling the Versatile Potentials of Cuprous Oxide: A Journey Through Material Science and Beyond
- Unveiling the Versatile Versatility of Copper Oxide: A Journey Through Science and Applications
- Silicone Marvels: Unveiling the Versatile Brilliance of Lithium Silicate
- Nano-cuprous oxide and nano-cupric oxide: similarities and differences in application fields
- Specific application process of concrete high-efficiency water reducing agent PCE powder in concrete
- The extraordinary journey of chromium oxide green in refractory materials
- Tungsten Oxide and Tungsten Trioxide: Unique Properties and Diverse Applications
- Know more about the Magnetite Black Iron Oxide - Fe3O4

